DIETARY SUPPLEMENTS
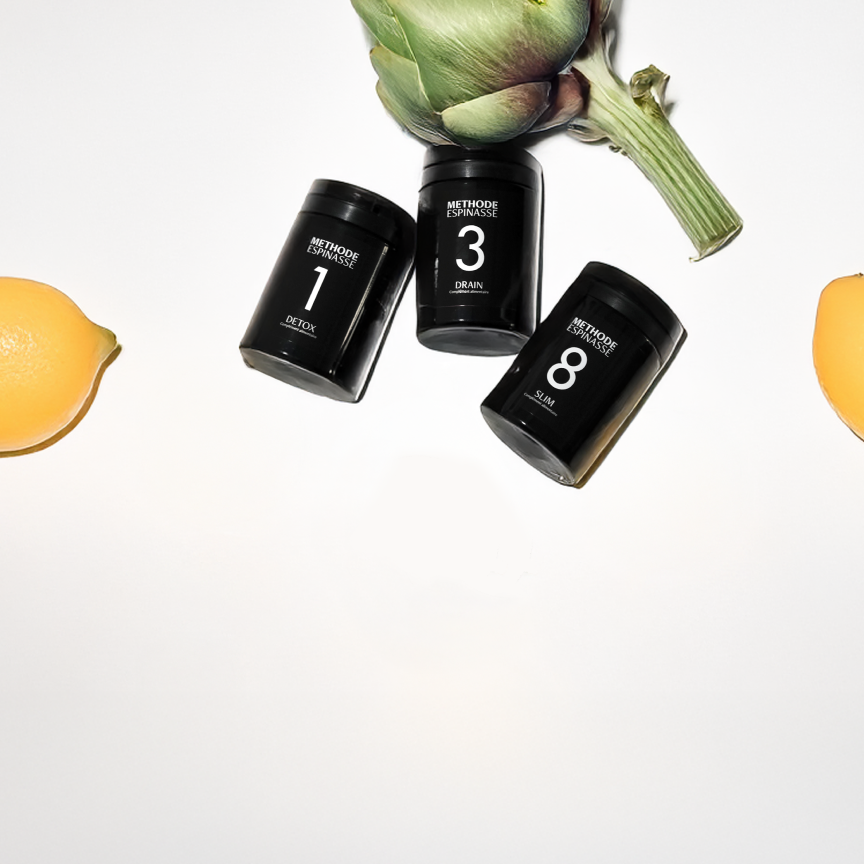
Découvrez 08 - Slim
Your ally for a slimmer figure and healthy weight

Journal
For decades, nutrition has been approached in relatively simple terms: meeting basic needs, avoiding deficiencies, balancing macronutrients. While this framework remains useful, it is no longer sufficient to explain what truly drives energy, recovery, aging, chronic inflammation, or metabolic resilience.
Modern research shows that health does not primarily operate at the level of the plate. It operates at the level of the cell [2].
This is precisely where cellular nutrition® comes in. Far from being a marketing concept, it reflects a more rigorous understanding of nutrition—not as the sum of isolated nutrients, but as a system capable of influencing the core biological networks that regulate cellular function.
These include mitochondria, metabolic signaling pathways such as mTOR and AMPK, autophagy, the gut microbiome, oxidative stress, and low-grade chronic inflammation [3][4].
In other words, eating well is not simply about “balanced meals.” It is about creating, day after day, a biological environment that supports optimal cellular function. This framework becomes essential when addressing longevity, prevention, metabolic health, chronic fatigue, weight regulation, skin aging, and cognitive performance [2].
Cellular nutrition® can be defined as a nutritional approach aimed at optimizing cellular function by acting on the biological mechanisms that regulate energy production, inflammation, stress response, cellular renewal, and metabolic adaptation.
This definition aligns with what the scientific literature now identifies as the key determinants of biological aging and healthspan [2].
The critical point is this: a cell does not respond solely to calorie intake or the theoretical presence of nutrients. It responds to signals.
These signals are nutritional, hormonal, inflammatory, microbial, oxidative, and environmental. Nutrient sensing pathways, in particular, translate nutritional status into cellular responses: growth, repair, storage, autophagy, energy production, or adaptation to stress [4].
This fundamentally changes how we think about nutrition. Two individuals may consume the same caloric intake yet experience completely different biological outcomes, depending on their microbiome composition, insulin sensitivity, inflammatory status, sleep quality, physical activity, micronutrient levels, and mitochondrial function.
Cellular nutrition® focuses precisely on this gap between what is consumed and what is actually processed, interpreted, and utilized at the cellular level [7].
One of the most important breakthroughs in aging research is the identification of the Hallmarks of Aging.
In their updated 2023 framework, these hallmarks include dysregulated nutrient sensing, mitochondrial dysfunction, chronic inflammation, dysbiosis, and impaired autophagy.
This is a critical point: the mechanisms targeted by cellular nutrition® are not peripheral—they are now recognized as central drivers of biological aging [2].
This does not mean nutrition “cures” aging—that would be scientifically inaccurate. However, it does mean that the nutritional environment can influence key pathways that accelerate or slow functional decline over time.
This marks the shift from a passive view of aging to a dynamic one, where biological processes remain, at least in part, modifiable [1].
Human data supports this perspective. In the CALERIE trial, moderate caloric restriction was associated with a measurable slowing of the pace of aging, assessed using the DunedinPACE algorithm, even if effects on other epigenetic clocks were more modest [11].
The magnitude of the effect remains moderate—but it reinforces the idea that nutritional interventions can influence biological aging trajectories.
If cellular nutrition® had to be summarized in one biological concept, it would be nutrient sensing.
Cells are equipped with systems that detect energy availability, amino acids, glucose, and other metabolic signals. Among the key regulators are mTOR and AMPK.
mTOR is broadly associated with growth and anabolic processes in conditions of nutrient abundance, while AMPK is activated under energy deficit and promotes metabolic adaptation [4].
This distinction should not be oversimplified. mTOR is not “bad,” and AMPK is not “good.” Both are essential. The issue arises when balance is chronically disrupted.
Persistent caloric excess, low metabolic flexibility, continuous nutrient stimulation, physical inactivity, mitochondrial dysfunction, poor sleep, and chronic inflammation can all shift these pathways in unfavorable directions [3].
This is why cellular nutrition® cannot be reduced to single ingredients. It depends on the overall quality of the nutritional signal delivered to the body: micronutrient density, protein quality, glycemic load, food matrix, fermentable fibers, polyphenols, meal timing, gut health, and physical activity.
All of these factors influence how cells decide between growth, repair, storage, and cellular cleanup [14].
No discussion of cellular health is complete without addressing mitochondria.
Mitochondria are not only responsible for ATP production—they also play a central role in redox signaling, stress adaptation, immune regulation, and inflammation.
With aging, mitochondrial efficiency tends to decline, leading to reduced energy production, increased oxidative stress, and diminished cellular resilience [3].
Recent research highlights the close interplay between mitochondria and nutrient sensing pathways. Mitochondria are not passive energy generators; they actively respond to nutritional status, physical activity, redox balance, and inflammatory signals.
When this regulation becomes impaired, fatigue, reduced physical capacity, metabolic dysfunction, and age-related decline become more likely [3].
This is also why exercise remains one of the most powerful interventions for mitochondrial health. Studies consistently show that training—even in older adults—enhances mitochondrial biogenesis, oxidative capacity, and overall function.
Any credible cellular nutrition® strategy must therefore integrate movement as a fundamental biological signal [15].
Another key dimension of cellular nutrition® is the management of inflammaging—the low-grade, chronic inflammation that develops over time and is now recognized as a major contributor to age-related diseases.
This process involves multiple mechanisms: cellular senescence, oxidative stress, mitochondrial dysfunction, persistent immune activation, increased intestinal permeability, and microbiome alterations [5].
This type of inflammation is often silent, diffuse, and long-lasting. Yet it profoundly alters cellular signaling, impairs insulin sensitivity, reduces recovery capacity, promotes tissue damage, and contributes to biological fatigue [5].
Cellular nutrition® aims to reduce this inflammatory background by acting on multiple levers simultaneously: dietary quality, polyphenol intake, glycemic control, micronutrient status, microbiome diversity, visceral fat regulation, and sleep quality.
Current research on Mediterranean dietary patterns and polyphenols supports their role in modulating inflammation, gut health, and healthy aging [13][14].
Cells do not operate in isolation from the intestinal environment.
The gut microbiome plays a crucial role in transforming dietary compounds, producing bioactive metabolites, modulating immune responses, maintaining gut barrier integrity, and communicating with the brain, muscles, and immune system.
Recent research even identifies dysbiosis as one of the hallmarks of aging [2].
This has major practical implications: a theoretically optimal diet may produce limited benefits if the gut ecosystem is impaired. Conversely, improving gut health can significantly alter nutrient absorption, metabolism, and systemic effects [9].
Scientific precision remains essential. Not all studies show uniform or dramatic changes in microbiome composition across dietary patterns. Even for well-established models like the Mediterranean diet, results can vary.
However, the overall evidence is clear: the diet–microbiome–inflammation axis is a central driver of health [7].
Cellular nutrition® is not only about providing nutrients—it is also about enabling cells to repair, recycle, and renew.
This is the role of autophagy, a cellular “housekeeping” process responsible for removing damaged components and maintaining cellular homeostasis.
Research shows that autophagy declines with age and is influenced by metabolic status, fasting, exercise, and nutrient sensing pathways [10].
This explains why effective cellular nutrition® is not about “more nutrients.” Constant anabolic stimulation, continuous energy availability, lack of movement, and poor metabolic flexibility can all impair the cell’s ability to enter efficient repair and renewal phases [4].
Two extremes should be avoided.
The first is reducing nutrition to generic dietary advice without biological depth. The second is claiming that nutrition can “reverse aging.”
The scientific evidence does not support such claims.
However, it does support the idea that certain nutritional and lifestyle environments can improve cellular resilience, slow some markers of biological aging, enhance mitochondrial function, reduce inflammation, and support metabolic health [11].
Cellular nutrition® operates within this realistic framework: not eliminating aging, but influencing the processes that make it faster or slower, more or less inflammatory, more or less functional [2].
A coherent cellular nutrition® strategy is not about multiplying supplements—it is about structuring a favorable biological environment.
In practice, this means a nutrient-dense diet rich in plant foods, fibers, polyphenols, and high-quality proteins, combined with better glycemic control and reduced intake of ultra-processed foods.
It also includes preserving muscle mass, optimizing gut health, maintaining regular physical activity, prioritizing sleep, and reducing chronic inflammation [13].
Supplementation can play a role—but only as an extension of a broader biological strategy.
Recent research on micronutrients and bioactive compounds suggests potential mechanistic effects on aging pathways, but outcomes depend heavily on context, baseline status, bioavailability, dosage, duration, and individual variability.
Once again, the key principle of cellular nutrition® is synergy—not accumulation [17].
The growing body of research on longevity, the microbiome, mitochondrial function, and precision nutrition reflects a broader shift in preventive medicine.
The focus is moving away from symptom management toward a deeper understanding of biological systems.
Cellular nutrition® fits squarely within this paradigm. It reframes the question—not “what should I take?” but “which biological systems need to be supported, in which context, and for what outcome?” [2].
This is likely why the concept resonates across fields such as longevity, metabolic health, performance, skin health, energy optimization, and preventive medicine.
It aligns with what modern biology clearly demonstrates: the human body functions as an interconnected network—not as isolated systems [2].
Cellular nutrition® is not a trend. It is a more accurate and biologically grounded way of understanding nutrition.
Scientific literature consistently shows that cellular health depends on a complex balance between energy production, metabolic signaling, inflammation, microbiome function, oxidative stress, and repair capacity.
Once this is understood, nutrition takes on a different role: it is no longer just about feeding the body—it becomes a tool to shape biology itself [2].
And this is precisely where the real value of cellular nutrition® lies: acting upstream, more deeply, and with far greater biological coherence than the traditional “one ingredient, one effect” approach [3].
[1] López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G.
The Hallmarks of Aging. Cell. 2013.
https://pubmed.ncbi.nlm.nih.gov/23746838/
[2] López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G.
Hallmarks of Aging: An Expanding Universe. Cell. 2023.
https://pubmed.ncbi.nlm.nih.gov/36599349/
[3] Tomteelnganbee E, et al.
Mitochondrial Function, Metabolism, and Nutrient Sensing in Aging. Ageing Research Reviews. 2022.
https://pubmed.ncbi.nlm.nih.gov/35842501/
[4] Saxton RA, Sabatini DM.
mTOR Signaling in Growth, Metabolism, and Disease. Cell. 2017.
https://pubmed.ncbi.nlm.nih.gov/28283069/
[5] Ferrucci L, Fabbri E.
Chronic Inflammation in Ageing, Cardiovascular Disease, and Frailty. Nature Reviews Cardiology. 2018.
https://pubmed.ncbi.nlm.nih.gov/30065258/
[6] Baechle JJ, et al.
Chronic Inflammation and the Hallmarks of Aging. Molecular Metabolism. 2023.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC10359950/
[7] Badal VD, Vaccariello ED, Murray ER, et al.
The Gut Microbiome, Aging, and Longevity: A Systematic Review. Nutrients. 2020.
https://pubmed.ncbi.nlm.nih.gov/33297486/
[8] Van Hul M, et al.
What Defines a Healthy Gut Microbiome? Nature Reviews Gastroenterology & Hepatology. 2024.
https://pubmed.ncbi.nlm.nih.gov/39322314/
[9] Tamayo M, et al.
How Diet and Lifestyle Can Fine-Tune the Gut Microbiome for Healthy Aging. 2024.
https://pubmed.ncbi.nlm.nih.gov/38941492/
[10] Raza S, et al.
Autophagy and Metabolic Aging: Current Understanding and Future Perspectives. 2024.
https://pubmed.ncbi.nlm.nih.gov/38763302/
[11] Waziry R, et al.
Effect of Long-Term Caloric Restriction on DNA Methylation Measures of Biological Aging (CALERIE Trial). Nature Aging. 2023.
https://pubmed.ncbi.nlm.nih.gov/37118425/
[12] Belsky DW, et al.
Quantification of the Pace of Biological Aging in Humans (DunedinPACE). eLife. 2020.
https://pubmed.ncbi.nlm.nih.gov/32367804/
[13] Ecarnot F, et al.
The Impact of the Mediterranean Diet on Immune Function and Healthy Aging. 2024.
https://pubmed.ncbi.nlm.nih.gov/38780713/
[14] Ross FC, et al.
Dietary Polyphenols and Protection from Age-Related Decline. 2024.
https://pubmed.ncbi.nlm.nih.gov/38287652/
[15] Memme JM, et al.
Exercise and Mitochondrial Health. Journal of Physiology. 2021.
https://pubmed.ncbi.nlm.nih.gov/31674658/
[16] Rinott E, et al.
The Effect of the Green Mediterranean Diet on Cardiometabolic Health and Gut Microbiome. Genome Medicine. 2022.
https://pubmed.ncbi.nlm.nih.gov/35264213/
[17] Dominguez LJ, et al.
Magnesium and the Hallmarks of Aging. 2024.
https://pubmed.ncbi.nlm.nih.gov/38398820/
[18] Kuerec AH, et al.
Targeting Aging with Urolithin A in Humans: A Systematic Review. 2024.
https://pubmed.ncbi.nlm.nih.gov/39002645/